Medtronic Linq II
The Medtronic Linq II is a project completed as part of a 30-week graduate course. Our team of interdisciplary graduate students was sponsored and guided by our client, Medtronic. We worked with Medtronic to develop a concept and business model for a device/digital application to improve pregnancy outcomes for those affected by preeclampsia.
Due to confidentiality agreements, portions of this project including the internal technology, mechanical prototype, financial modeling, and user research, have been removed from this presentation.
Industry: Medical Device
Role: UX Designer, CAD modeling, User-Research
Duration: 30 weeks
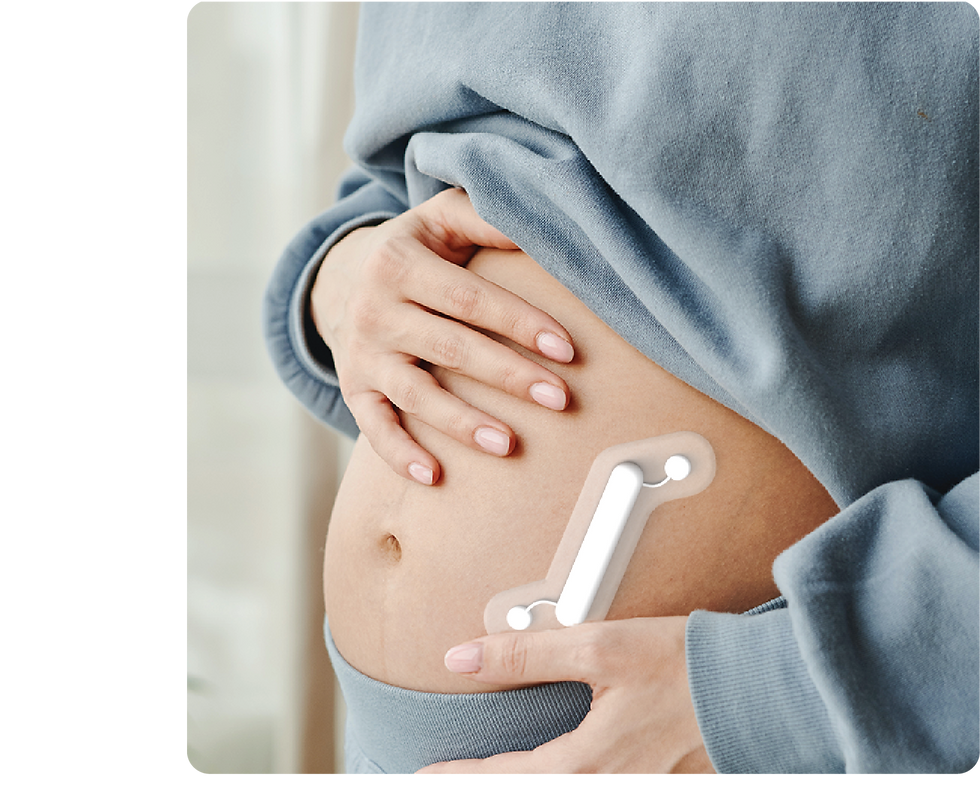


Uncovering the Problem
Our team began by examining the whole pregnancy journey and conducting a market analysis to identify areas of opportunity. This initial research resulted in defining our scope to high risk pregnancies caused by preeclampsia.
What is preeclampsia?
Preeclampsia is a serious condition that can occur during pregnancy or after giving birth, and is characterized by high blood pressure and other signs of organ damage.
Who is affected?
Preeclampsia affects approximately 3 to 6 percent of pregnancies per year (U.S), with that risk being up to twice as high for those experiencing their first pregnancies (Anath et al., 2013).
Market Opportunity
Market research revealed that there are few self-monitoring products that are specific to preeclampsia. Current solutions include blood pressure cuffs and personal doplers, though these products are not always accurate and can cause unnecessary worry for patients. Another solution for care teams is to have the patient stay in-hospital for surveillance.
Project Goals
The goal of this project was to design a concept for an interactive patient monitoring system that enables healthcare providers to efficiently track and manage patient and fetal health data. This system should enhance the workflow of medical professionals, ease patient minds, and decrease unnecessary ER visits during the pregnancy experience.

User Research
Our team sourced patient paticipants through our own social circles. We sourced providers (OBGYNs) through M Health Fairview. My role in user research was standardize patient-facing interviews and interpret participant data. Together we completed 40 patient interviews and 10 physician internviews.

Physicians:
7 OBGYNs, 3 Cardiologists
What do we want to learn?
-
How do phsyicians currently detect preeclampsia?
-
Feedback on low-fi concept sketches.

Patients:
40 women who had experienced preeclampsia during pregnancy
What do we want to learn?
-
Attitudes reguarding patient montoring.
-
Feedback on low-fi concept sketches.
Synthesizing data from user research
Of particular value to our project was analyzing qualtative findings. In the example above, I took key "moments" from interviewers that captured their attitudes reguarding wearble vs implantable technology. This graphic provided evidence to the team and our Medtronic partners that the majority of women favored wearable monitoring. These and other finding shaped our concept development.

Of particular value to our project was analyzing qualtative findings. In the example above, I took key "moments" from interviewers that captured their attitudes reguarding wearble vs implantable technology. This graphic provided evidence to the team and our Medtronic partners that the majority of women favored wearable monitoring. These and other findings shaped our concept development for both the physical and digital products.

Digital platform development
Based on these findings, we developed two prototypes in Figma, patient-facing interface and a provider-facing interface. Since the needs of these user groups differed, the prototypes included their own freatures. For example, our team focused customization features for patients while the provider interface required a feature to link the physical device with the patient's account. Both user groups required their own set of data visualization features.
1. Patient-facing user interface
-
Allow real-time vital monitoring, yet remain data sensitive.
-
Provide confirmation when data is successfully reported to care team.
-
Develop a notification protocol to avoid unnessesary panick and the ability for consumers to customize how they would like to be contacted.
2. Provider-facing user interface
-
Ability for care team to request a data transmit.
-
Communicate "long data" so it can be interpreted for patterns/trends.
Conclusion and Future Work
Our user research findings and prototypes were compiled into a report and into a presentation format in which our prototypes were pitched to a Medtronic forum. We were given valuable feedback to improve both the product as well as the user experience.
If given the opportunity, I would advocate for more extensive testing of the device and platform to ensure accuracy, reliability, and user-friendliness across diverse populations. Our user pool was limited our own social networks. Increasing our sample size of users would ensure that the device meets high standards of quality and efficacy.
I would also like to address the issue of the product lifecycle. The current trend towards disposable medical devices contributes significantly to waste generation in healthcare. Therefore, I would propose developing a program where the device, or parts of the device are reused to help reduce waste and mitigate price. It would be interesting to build a prototype of that experience.
